The Compliance Check feature is a paid feature available in a special tab in the Ingredient section. Supplier's Guide checks the compliance using 3rd party regulatory reference database which contains over 100 000 regulations from over 200 countries. The database is updated monthly. Before you can start using the feature, you need to apply your unique token.
It is necessary to ensure correct configuration. Inaccurate configuration may lead to completely incorrect results - prohibited/restricted substances may show as permitted!
The ingredients needs to be mapped to the IDs of the ingredients in the reference database (DID). The mapping is either done in the SpecPDM and then uploaded to the Supplier's Guide via interface. Mapping of the ingredients functions is described in the SpecPDM manual. For customers not using the SpecPDM, the ingredients mapping can be imported by SpecPage. Additionally, the additive classes need to be mapped to the ingredients functions or you use can use the default mapping. The function determines the reason why the food additive is being utilized. The meaning is similar to the meaning of additive class (class antioxidant = function antioxidant), but it is not always the case. If there is no additive class selected by the supplier, or if the mapping between the class and function is missing, then the result will be shown for all functions.
The regulatory compliance check is done by the issuer in the Ingredients section for the:
▪ingredients entered by the supplier
▪additive classes entered by the supplier
▪countries selected by the issuer (mandatory)
▪usage selected by the issuer (optional)
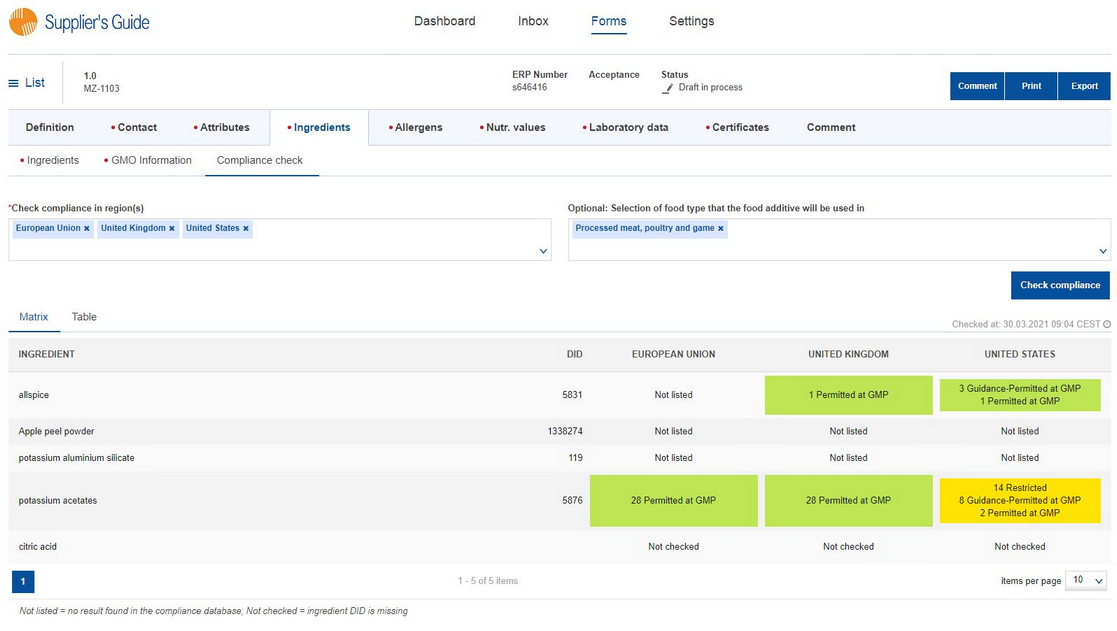
In the Ingredients section, go to the Compliance check tab. Select the countries in which you want to check the compliance, optionally select the usage(s) and request the regulatory check results by clicking on the "Check compliance" button.
The results are displayed in two views: Matrix and Table. The Matrix view represents a summary of the results. It provides you with information about the settings you applied and the results for all the countries you selected side by side. The Table view lets you see more detailed information including the existing thresholds and the corresponding regulations. Click on the citation, if available, to open the regulation in your default web browser.
The results of the compliance check are saved unless the specification is accepted, archived or deleted. You can see the time-stamp of the last check. You can do the check after the specification is accepted and the results will be displayed to you but the results will not be saved. The reason is to store the historical results that were valid before the specification was accepted so that it is possible to check what was the regulatory compliance at that time.
Result Indicator |
Definition |
Prohibited |
Not allowable per regulation or banned substance list |
Required |
There is a minimum threshold limit in place by regulation; sometimes there is also a maximum threshold limit |
Restricted |
There is a maximum threshold limit in place by regulation |
Recommended |
This applies to voluntary standards where there is a minimum threshold limit in place by regulation; sometimes there is also a maximum threshold limit. |
Permitted at GMP |
Allowed to be used as a food additive. The threshold established or required by regulation is Good Manufacturing Practices (GMP) or quantum satis (QS). |
Prohibited if no limit applies |
This applies when there is a list of substances permitted for foodstuffs, but there is a second table where the substance is prohibited in certain usages unless there is specific data elsewhere in the regulation that allows the use of this substance with a maximum threshold limit. |
Guidance-Prohibited |
This applies when the prohibition or banned results comes from a guidance or advisory document rather than a regulation or legal requirement. |
Guidance-Restricted |
This applies when the restriction comes from a guidance or advisory document rather than a regulation or legal requirement. |
Guidance-Permitted at GMP |
This applies when the permission comes from a guidance or advisory document rather than a regulation or legal requirement. |
Listed |
This applies when there is not enough information present in the regulation to make a definite decision about the substance found in the regulation. These usually require further evaluation by customers. |
Guidance-Listed |
This applies when there is not enough information present in the guidance document to make a definite decision about a threshold. These usually require further evaluation by customers. |
There might be no results for the ingredients provided by the supplier. In that case the result shows one of the following states.
▪Not listed = no result found in the compliance database;
▪Not checked = ingredient DID is missing;
Suppliers can see the results of the compliance check that the issuer did. The Compliance check tab is visible also for the suppliers, but suppliers cannot do the checks on their own. The selection of countries, usages is disable and the button to do the compliance check is missing.